If you’re a dermatologist engaged in clinical research, you may be looking for tips on optimizing the regulatory process for your CRO. For instance, you might be concerned that the raters you work with need to be qualified enough to carry out rigorous testing and data monitoring. You also might be worried that your CRO needs to put the patient’s needs at the center of the research process.
Challenges with Dermatology CROs
Dermatology CROs are a critical component of drug development. However, there are some challenges facing the industry. These include commoditization, increased competition, and regulatory requirements. Moreover, the cost of dermatology CROs is on the rise. As dermatology and aesthetic medicine are increasing, pharmaceutical companies must improve their research services. In addition, government initiatives to promote efficiency in the healthcare supply chain are expected to drive market growth. The most significant trends in the dermatology CROs market are the adoption of advanced software solutions and the integration of advanced trial programs. This is a result of increased R&D activities. Moreover, the aging population and rising awareness of skin ailments are also predicted to boost the market. Another major factor driving the dermatology CRO market is the increased demand for biologics and regenerative medicines. A growing need for topical dermatological medications such as cleansers, emollients, anti-infectives, and local anesthetics is also expected to drive the industry.
In addition to providing a wide range of dermatology services, dermatology CROs offer quick turnaround times. Additionally, they ensure that clinical trials are carried out according to good clinical practice. Therefore, dermatology CROs are highly sought after by biotech companies. Increased government support for R&D activities is also a significant factor supporting the industry’s growth. Furthermore, government initiatives to streamline the supply chain are also predicted to boost the dermatology CROs market. Another major trend in the dermatology CROs market is the integration of machine learning platforms. Machine learning is a process that allows CROs to make decisions about skin disorders, treatments, and product development.
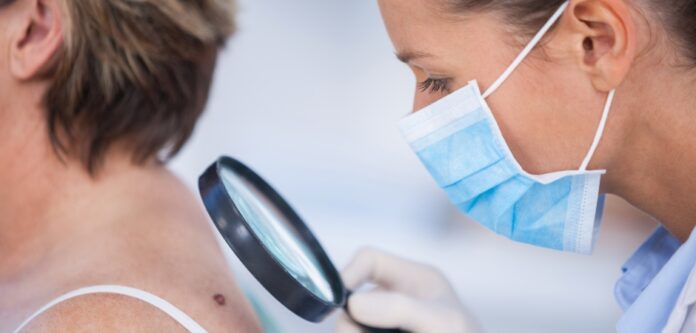
Developing and rolling out rigorous rater training, testing, and data monitoring
In addition to providing high-quality data, reliable rater training, testing, and data monitoring are critical to the success of dermatology studies. Sponsors must ensure that their studies adhere to the protocol and that their rates are consistent. These are all essential for downstream regulatory submissions and to ensure a successful market launch. The shift toward more decentralized trial designs will improve patient, physician, and sponsor outcomes. By addressing specific patient needs, studies will be more efficient, which means reduced study timelines and resource usage.
Dermatology clinical trial patients typically seek improved skin conditions and quality of life. They also may be motivated to participate in a trial by a desire to contribute to the body of knowledge. Because of this, sponsors may perceive a tension between the priority of clinical trials and the importance of patients. A patient-focused CRO can help ease this tension by making studies more accessible and allowing for an increased participation rate.
Working with an established CRO network can ease the start-up process. This way, studies are streamlined, and paperwork can be reduced. When working with a dermatology-focused CRO, the analysis can differentiate itself from similar trials and provide a fantastic opportunity for market adoption. Using a patient-focused approach can also increase study visibility and accelerate the path to commercialization. For example, a focus group or patient advisory board can help establish ongoing conversations. Moreover, targeted site visits can inform remedial training for investigators.
Multipronged EHR optimization approaches for Dermatology CROs
When optimizing your EHR, multipronged approaches are the way to go. The key to success is to implement appropriate engagement with key stakeholders. These include clinicians and administrative staff. You can reap the rewards of a well-implemented solution by putting them on the same page. One of the essential facets of this undertaking is selecting a well-vetted EHR system. Your choice should be based on the care your organization provides and its current infrastructure and capabilities. However, you must also consider how you will utilize and optimize this new technology to deliver the best patient care possible. There are many ways to achieve this. For instance, EHR software packages can automatically import images and audiogram reports into the electronic medical record. Other solutions, such as visual documentation engines, can generate exam documentation based on your diagnostic and treatment plans.
Another option is to use an existing condition-specific template to create a standardized framework for exchanging clinical data between your PCP and specialist. While these templates have shortcomings, they can facilitate communication between the two parties. An intelligent algorithm is needed to optimize the flow of information and the cost of doing so.
While there are many options to choose from, it is crucial to understand your organization’s unique needs and constraints. A good understanding of your physicians’ daily routines and workflows will help you tailor the solution to your specific challenges. In particular, you may need to make changes to the type and size of the database, as well as its structure and content. It’s essential to be aware that changes in the EHR will often coincide with changes in the broader organizational policy landscape. Your strategy may need to take the form of several more minor, targeted interventions. Finally, it’s vital to recognize that implementing technological advancements requires a healthy strategic plan. Your leadership should look closely at how the EHR is currently being used to ensure it works to its fullest potential.









![Anso FG Reviews: UPDATED 2024 [ansofg.com] Anso FG Reviews UPDATED 2024 [ansofg.com]](/wp-content/uploads/2023/12/Anso-FG-Reviews-UPDATED-2024-ansofg.com_-100x70.png)







